Consistently Ranked Among the Top 3 in Digital Pathology
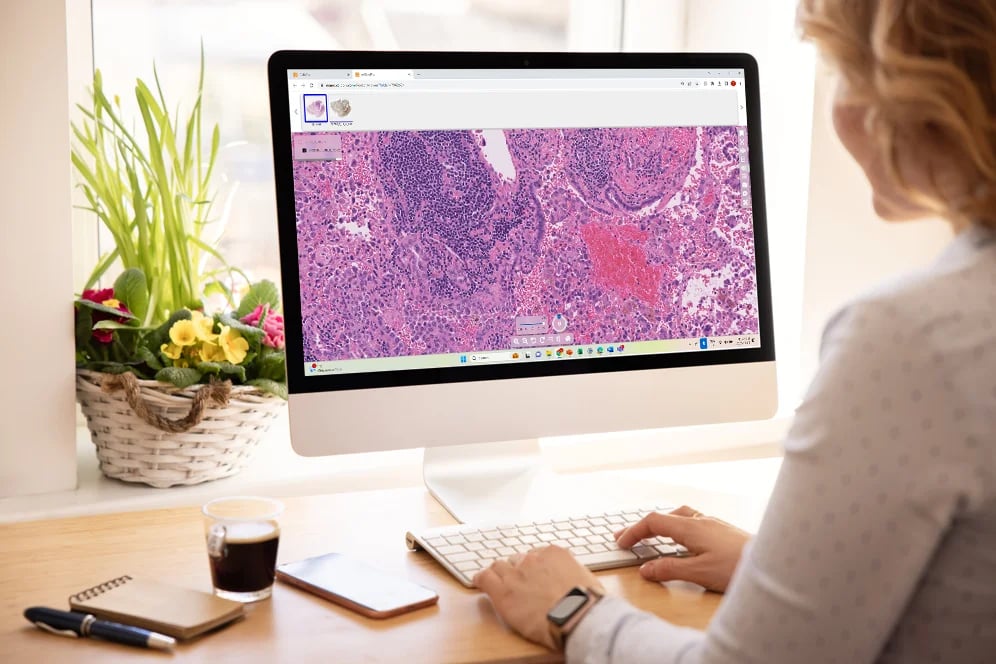
Healthcare organizations adopting digital pathology need technology that performs in real clinical environments—not in theory. CaloPix®, Tribun Health’s Image Management System (IMS), supports hospitals transitioning from microscope-based workflows to fully digital diagnostic practice, with deployments across academic centers, regional networks, and national health systems.
Recognized by KLAS Research as Best in KLAS for Digital Pathology for three consecutive years (2022–2024), CaloPix® continues to rank among the top 3 solutions in 2025 and 2026—reflecting sustained user satisfaction and proven performance in daily clinical use. Based entirely on feedback from healthcare providers, KLAS evaluations confirm the platform’s reliability, scalability, and value in supporting routine diagnosis, collaboration, and enterprise imaging strategies. As Jean-François Pomerol, CEO of Tribun Health, states: “Without the trust and evaluations of our customers, this recognition would not be possible. Our team is proud to contribute to advancing healthcare.”
Since 1996, KLAS Research has provided independent, unbiased insights into healthcare technology, with a mission to improve care worldwide by amplifying the voice of providers and users.





.png?width=256&height=256&name=customer-service(1).png)



.png?width=64&height=64&name=calendrier(1).png)
.png?width=64&height=64&name=communique-de-presse(1).png)
.png?width=64&height=64&name=livre(1).png)
.png?width=64&height=64&name=blog(2).png)